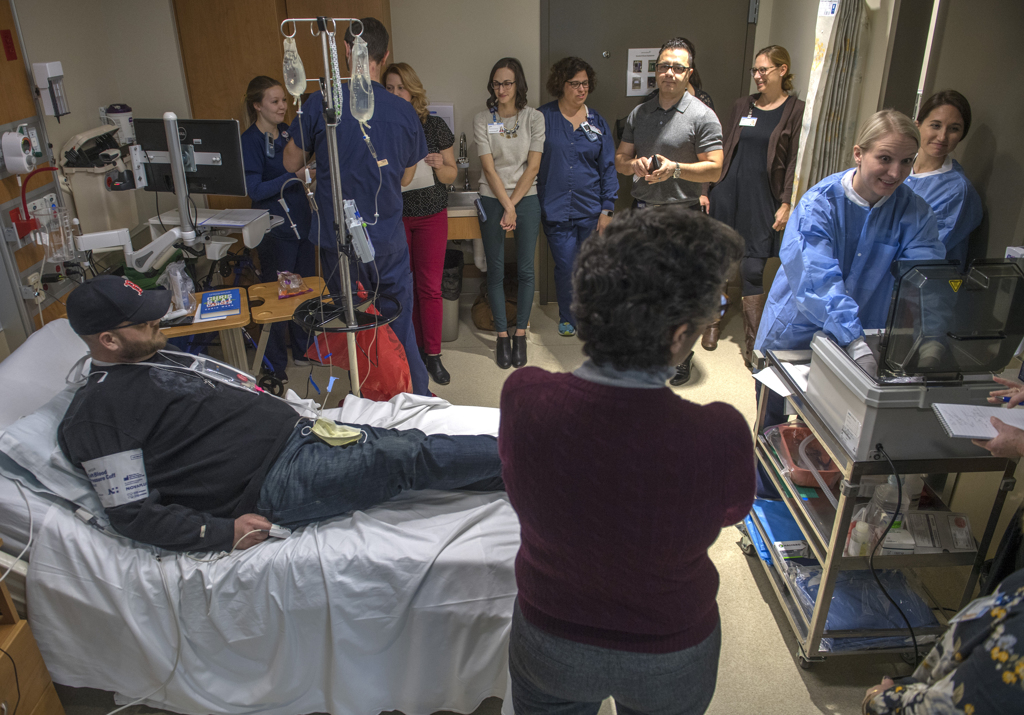
Jeremy Rootes raised his cellphone to take pictures as a nurse hung a thin, plastic bag of medicine on his intravenous pole.
In a six-year battle with lymphoma, Rootes had lived through many cancer treatments. He knew both the joy of remission and the panic of relapse.
But this was different, this foggy swirl of liquid.
In it lay millions of his own T-cells, re-engineered to seek out and destroy cancer cells that stubbornly outlasted the standard weapons in the anti-cancer arsenal.
Called CAR T-cell therapy, the groundbreaking treatment is a new and sophisticated form of immunotherapy.
“It’s exciting because it’s a totally different approach,” said Rootes, a 38-year-old salesman from Grandville, Michigan, who has non-Hodgkin lymphoma. “This directly attacks what my problem is, which is awesome.”
The U.S. Food and Drug Administration approved CAR T-cell therapy for two forms of cancer in 2017. More than 300 clinical trials in the U.S. are exploring whether it can be effective against other forms of cancer.
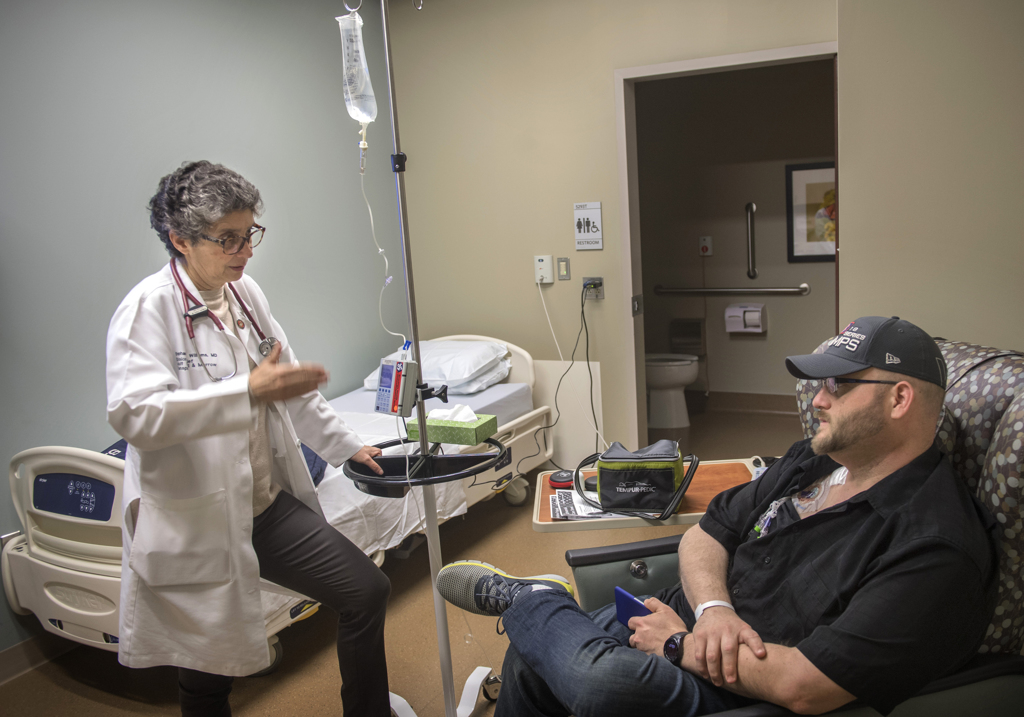
Rootes became the first person to receive the treatment through Spectrum Health Cancer Center’s adult blood and marrow transplant program when he received the infusion in December 2018.
“I think it’s a very exciting time,” said Stephanie Williams, MD, the division chief of Spectrum Health’s adult blood and marrow transplant program.
“It’s really a time where the paradigm of treating cancer is shifting. Hopefully, it’s shifting toward a more targeted, rational approach to killing cancer cells.”
It started with a lump
Rootes noticed the first signs of cancer eight years ago, when a growth appeared on his neck. Over the next two years, he underwent several needlepoint aspirations that found no sign of malignancy.
But still the lump grew. It impeded his breathing and swallowing. He contacted an otolaryngologist, who performed a biopsy. The results showed he had non-Hodgkin lymphoma, a cancer that starts in white blood cells called lymphocytes.
He was 32 years old. He had a 3-year-old son and a newborn daughter. And suddenly cancer treatments grabbed center stage in his busy life.
“Much, much chemo. A month of radiation. A stem cell transplant,” he said. “It was extensive.”
The cancer went into remission. But in November 2017, a routine PET scan showed the cancer was back. Rootes also had a tumor on his right kidney, unrelated to the lymphoma.
Again, cancer turned his life upside down.
“Every step of the way, it’s never not a shock,” he said.
But this time, he had a new source of hope.
It’s still early, but the response appears to be long lasting.
Just one month before his relapse, the FDA approved CAR T-cell therapy for diffuse large B-cell lymphoma—the same type of cancer Rootes has.
The approval came after a study found promising results. In patients with relapsed or refractory large B-cell lymphoma, 51 percent achieved complete remission.
It became just the second form of cancer for which the FDA approved CAR T-cell therapy. The agency had also approved the therapy for treating children and young adults with acute lymphoblastic leukemia.
Working with his oncologist, Rootes made plans to go out of state for the therapy—until he learned Spectrum Health planned to offer it in Grand Rapids, Michigan. He opted to have the treatment close to home, working with physicians recommended by his oncologist.
In January 2018, he underwent surgery to remove his kidney. And he began receiving treatment for lymphoma.
“He was getting therapy that was holding it at bay, but it was bound to come back,” Dr. Williams said. CAR T-cell therapy was “his best option, and probably his last hope, for treatment.”
In November, he took his first step in the therapy. Doctors collected his lymphocytes.
“You use the same machine as when someone is donating platelets,” said Ulrich Duffner, MD, a pediatric blood and marrow transplant specialist and the quality director for CAR T-cell therapy at Spectrum Health.
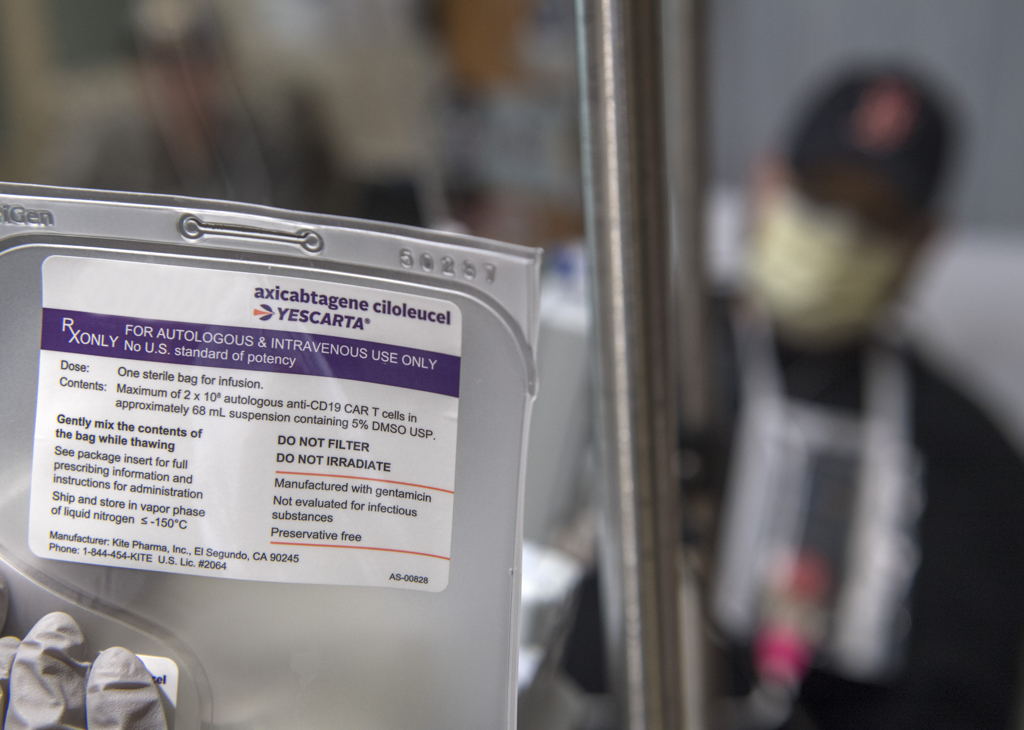
Blood drawn from the body goes through the machine, which pulls out T-cells—a type of immune system cell—and returns the blood back to the body. The T-cells are sent to Kite Pharma, the company in California that developed the therapy for lymphoma.
The company genetically modifies the cells so they include receptors called chimeric antigen receptors, or CAR. These special receptors recognize and bind to lymphoma cells and kill them.
“The receptor is connected to some very strong amplifiers,” Dr. Duffner added. “When the CAR T-cell gets signaling from the receptor, it starts proliferating. It tells the immune system to start being active and they start killing the leukemia (or lymphoma) cells.”
The modified T-cells multiply 1,000-fold, creating a major assault on the cancer cells.
Ready for battle
Three weeks after the T-cells were removed from Rootes’ blood, it was time for the next step. He approached it with mixed feelings.
“I’m excited, nervous and terrified in equal parts,” he said. The week before, he had gone through several days of chemo preparing for the infusion.
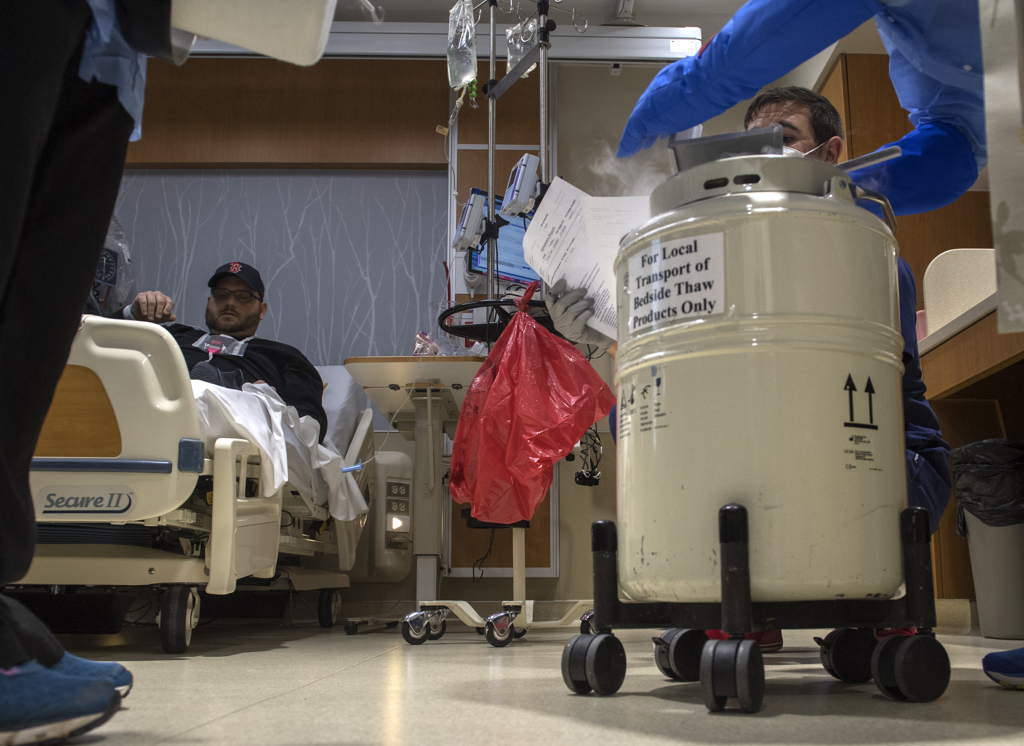
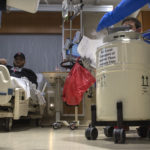
Rootes sat in a bed in the Lettinga Inpatient Cancer Center at Spectrum Health Butterworth Hospital as two Michigan Blood employees delivered a large metal canister.
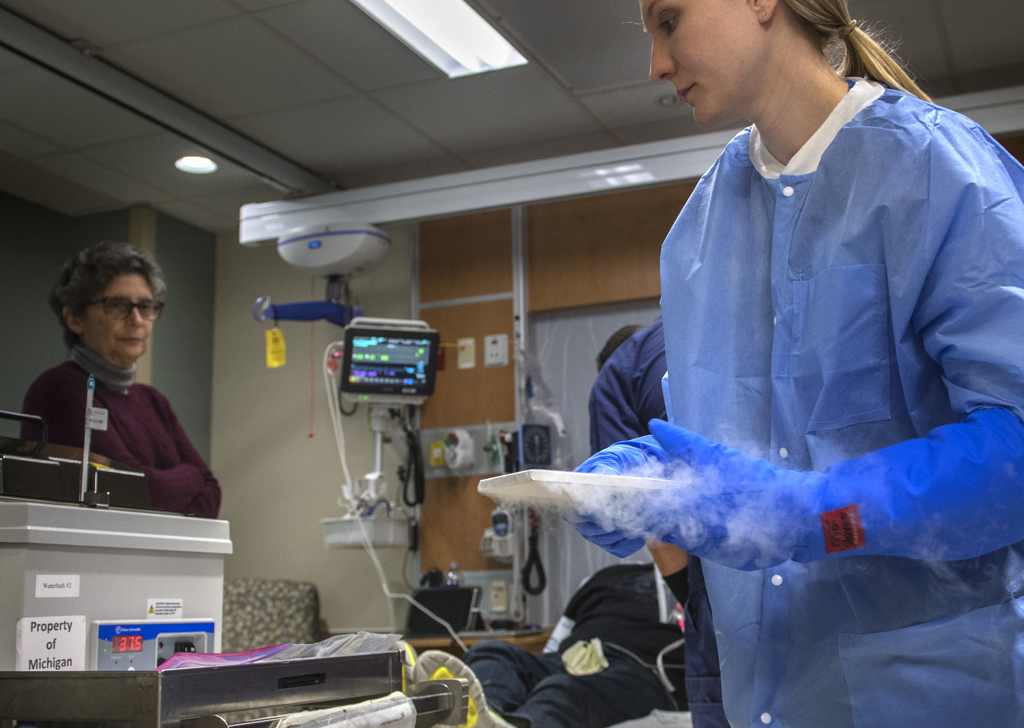
They removed the lid and a puff of white fog rose up. The canister kept the cells frozen at 180 degrees below zero.
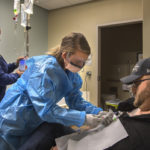
The workers wore heavy gloves as they withdrew a thin metal box, opened it and brought out the small plastic bag containing frozen T-cells. They thawed it in a basin of cool water.
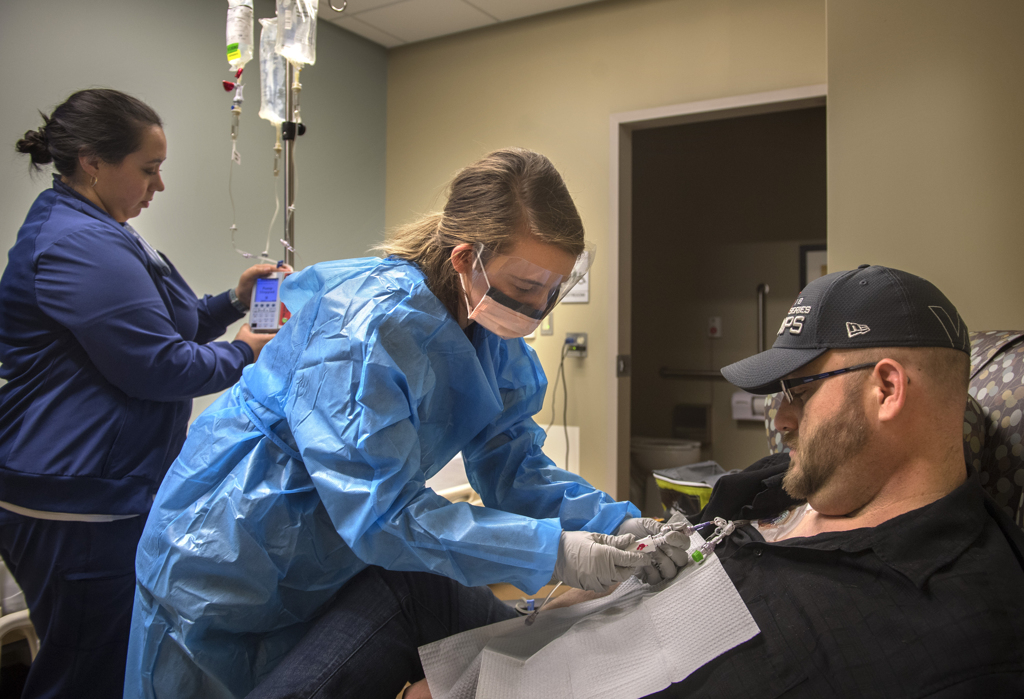
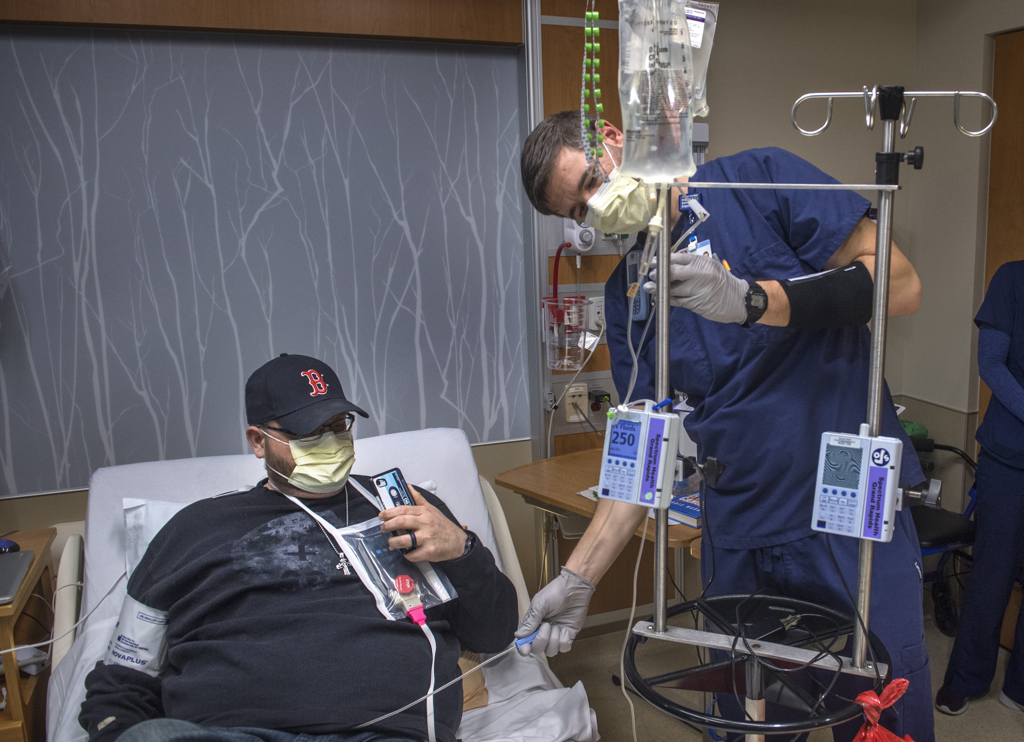
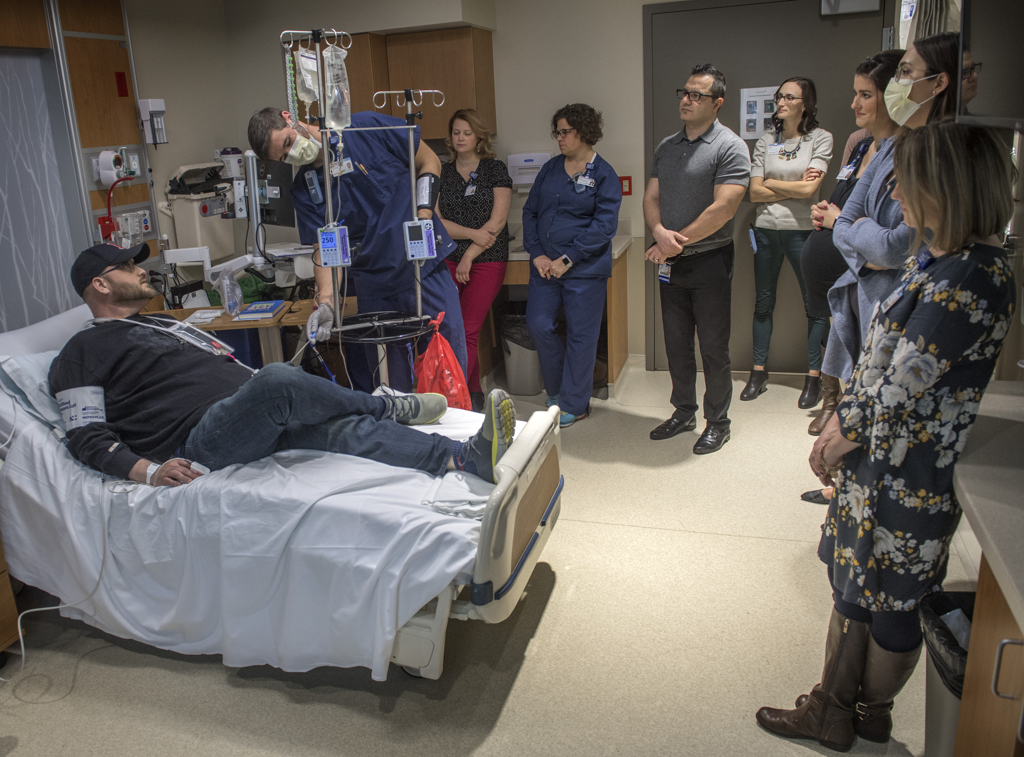
“This is a historic moment,” said Josh Hendricks, RN, the nurse on the blood and marrow transplant unit who helped prepare Rootes for the infusion.
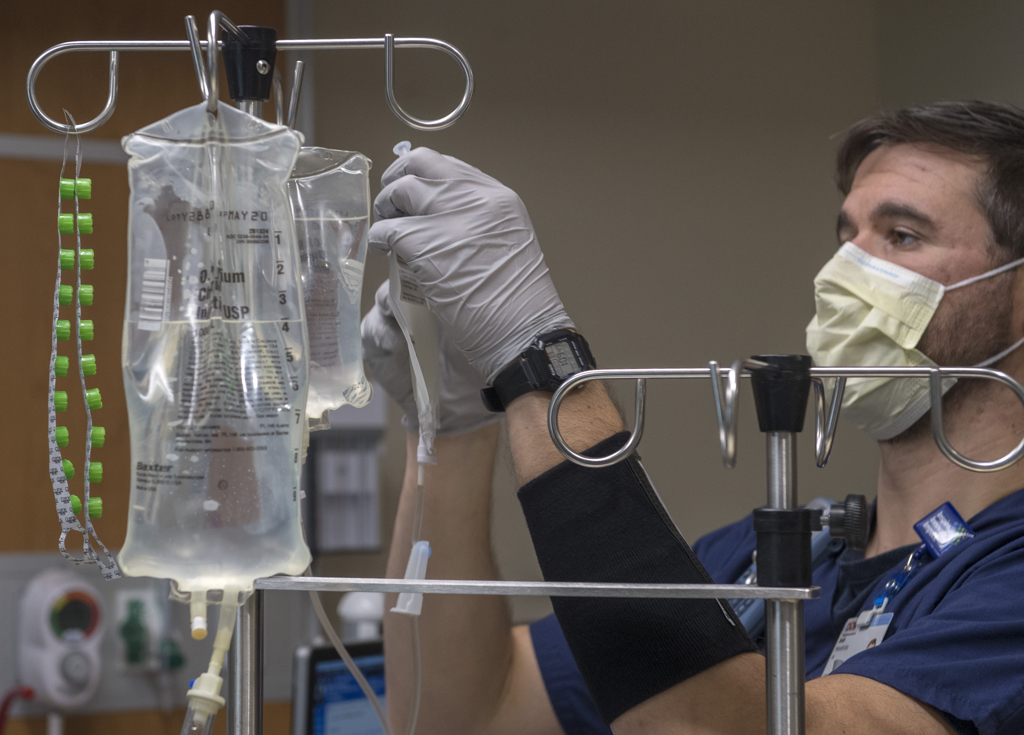
He hung the bag on the IV pole and connected it to the catheter already inserted in Rootes’ vein. The IV held a little more than 2 ounces of liquid.
Hendricks opened a valve and the liquid began to drip down the tube.
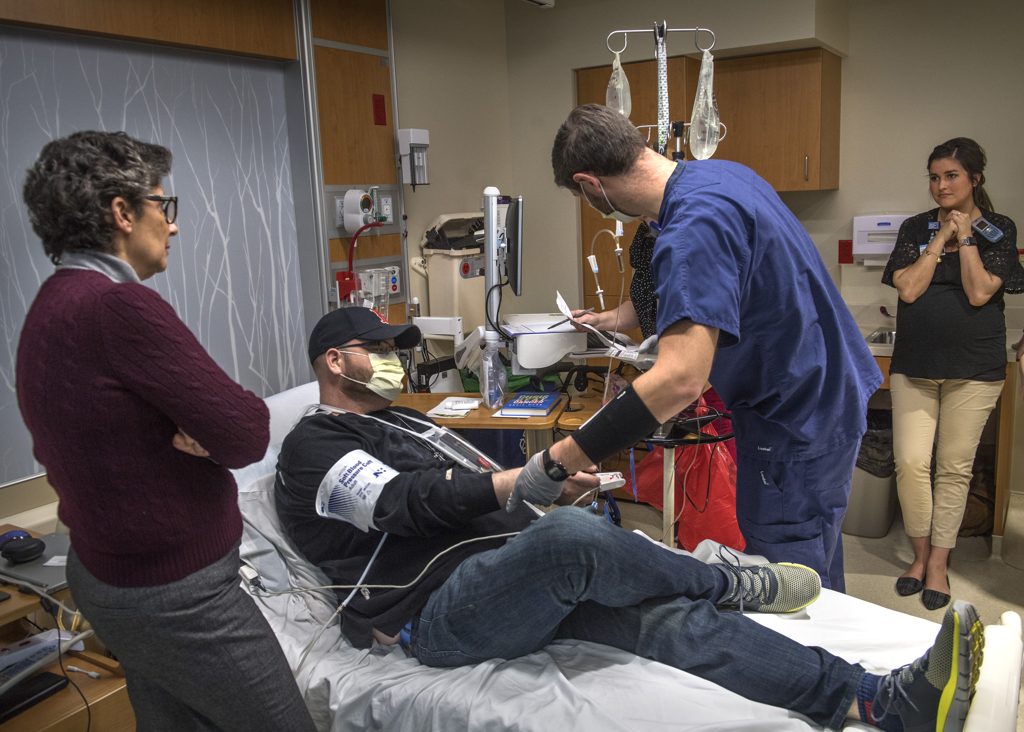
“Jeremy, remember it might be a little chilly,” Dr. Williams said.
He nodded. He had experienced that cold infusion during his stem cell transplant a few years ago.
The infusion also created an odd taste in his mouth, caused by the preservative. He felt as if he’d just eaten garlic or creamed corn.
Eleven minutes later, the last of the T-cells had been flushed from the bag and fed into Rootes’ vein. Chilled, he asked for an extra blanket.
Careful monitoring
The next steps included watching for side effects, which can be serious and even life-threatening. In ramping up the immune system, the therapy can cause high fevers, decreased blood pressure and neurological or cardiac issues. Patients can experience cytokine release syndrome, a systemic inflammatory response.
Before Spectrum Health began to offer CAR T-cell therapy, a multidisciplinary team prepared a detailed plan for recognizing, managing and treating the side effects.
“You need to monitor these patients around the clock,” Dr. Duffner said. “When somebody shows signs of one of these things, you need to have a very leveled response. The cytokine release syndrome is part of why these cells are so effective.”
Side effects usually surface around three days after transplant and can occur in the first two to four weeks. However, the side effects usually are temporary.
“CAR T-cell patients can get quite sick from the treatment, but as far as we know it’s a temporary thing,” Dr. Duffner said. “Once you have treated it, it’s done and it doesn’t come back.”
I think we have found the key we’ve been looking for a very long time.
Because the therapy is relatively new, researchers still are investigating its long-term impact. A key question is whether the modified T-cells will function like a “living drug,” remaining in the body for months or years, activating if new lymphoma cells show up.
“It’s still early, but the response appears to be long lasting,” Dr. Williams said. “We don’t know if it cures people yet.”
Researchers also are investigating whether CAR T-cell therapy can be effective in treating other forms of cancer, such as multiple myeloma, and solid tumors of the lung, brain, breast and colon.
Off to a good start
After the CAR T-cell infusion, Rootes met with Dr. Williams regularly for follow-up visits and blood work.
Nine days post-treatment, she said, “He’s doing great. We are very pleased.”
So far, Rootes said he avoided most of the possible side effects.
“I had one low-grade fever on Friday night (four days after the infusion) and I felt like crap all day Saturday,” he said. “I woke up Sunday feeling like a million bucks and that hasn’t gone away.”
Rootes is optimistic about the therapy—for himself and for treating other forms of cancer still under investigation.
“I think we have found the key we’ve been looking for a very long time,” he said.




















 /a>
/a>
 /a>
/a>
 /a>
/a>
What an amazing story and hope for more people. Thank you for sharing.
Thank you, Amy! We’re glad to have you as part of the Health Beat family. 🙂
Is this similar to plasma pheresis that my dad had for Guillan Barre Syndrome? Is CAR T-cell therapy used for CLL, too.
It’s a inspirational story which gives me hope as I am living with stage 3 multiple myeloma.
I’m glad you find it inspirational. Wishing you the best as you deal with your own cancer diagnosis.